  
The quest for high-intensity white LEDs gave rise to a red-green-blue mix that the eye perceives as white, with incrementally improved color rendition. Subsequent bright blue LED improvements made Blu-ray a reality. High-intensity blue LEDs emerged in 1991. Blue LEDs and laser diodes were too dim and gallium nitride devices were discontinued until, in 1989, a semiconductor manufacturer based in North Carolina introduced a commercially available but inefficient blue LED based on an indirect bandgap semiconductor, silicon carbide (SiC). But LED colors other than red and some other narrow-band hues remained elusive. In the 1970s, Fairchild Optoelectronics produced LEDs priced below five cents each, based on Atalla’s surface passivation technology. That changed when Mohamed Atalla, renowned MOSFET inventor, and his associates at Hewlett Packard collaborated to develop new LEDs using GaAs, GaAsP and InAs devices. Then, George Craford in 1972 demonstrated a yellow LED and greatly increased the intensity of red and orange LEDs.Įarly LED’s played a role in laboratory test equipment, but they were still too costly, in excess of $200 per unit, for general usage. Soon Texas Instruments began making infrared diodes and LEDs that used GaAs crystals emitting 890 nm light beams.Īt General Electric, Nick Holonyak build the world’s first visible red LED. Subsequently, they observed light emission as well as signal transmission between a GaAs P-N light source and a semiconducting photodetector. James Beard and Gary Pittman, working at Texas Instruments, placed a tunnel diode on a GaAs substrate, producing near-infrared emission. Additionally, he reported room-temperature infrared emission in diodes using gallium antimonide (GaSb), GaAs, indium phosphide (InP) and silicon germanium. In 1955, Rubin Braunstein in RCA Labs, observed infrared emission in gallium arsenide (GaAs) and other semiconductors. Georges Destriau observed electroluminescence when he applied an ac voltage to zinc sulfide (ZnS) powder suspended in an insulator. Two decades later, a Russian investigator, Oleg Losev created the first true LED. Actually, in 1907, Henry Joseph Round observed luminescence in a solid-state diode, using a cat’s whisker silicon carbide detector. You might think the idea for LEDs came along shortly before their commercial debut. Back then, LEDs weren’t entrusted to mere undergrad engineering students.) (Engineers who were in school during the early 1970s, when LEDs first became commercialized, can recall lab sessions where undergrads peered in at a dimly lit LED which was part of a circuit set up by lab instructors. Over the years they evolved, first becoming primitive indicator lamps and then seven-segment displays. The first LEDs were used in TV remotes, emitting a low-intensity, narrow infrared beam sufficient only to be sensed a few feet away. Many commercial LEDs also use a sapphire substrate. P-type substrates, while less common, ae used as well. LEDs are usually built on an n-type substrate, with an electrode attached to the p-type layer deposited on its surface. Another type of LED uses a quantum dot which can have its properties and wavelength adjusted by its size. In modern-day LEDs, materials such as gallium arsenide with various trace doping elements are used to produce different colors of light. Most of the light produced tends to reflect back from the surface of the semiconductor (high index of refraction), so LEDs use special optical coatings and die shapes to boost light emission. The wavelength of the light produced depends on the energy band gap of the LED semiconductors. The recombination of electrons and electron holes in the LED produce light via electroluminescence. If the semiconductor material is translucent, the diode junction becomes a source of light, thus creating a light-emitting diode. But in diodes composed of gallium arsenide phosphide (GaAsP) and gallium phosphide (GaP), the electrons dissipate energy by emitting photons. In ordinary silicon and germanium diodes, electrons flowing through the diode dissipate energy in the form of heat. LED charge carrier behavior as explained in Wikipedia. This energy is emitted in the form of heat and light. Some portion of the energy must be dissipated to recombine the electrons and the holes. 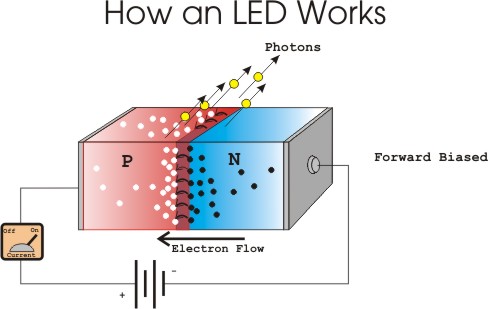
Thus the energy level of the holes is lower than the energy levels of the electrons. Free electrons are in the conduction band of semiconductor energy levels, while holes are in the valence energy band. Under a forward voltage bias, electrons cross from the n-region and recombine with the holes in the p-region. The LED is a diode that emits light only when forward-biased. The LED or light-emitting diode emits light while the photovoltaic cell converts light to electricity. 
LED’s and photovoltaic cells play increasingly prominent roles in our world. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
